Objective: To synthesize selected literature into a structured clinical framework for conservative crosslinked hyaluronic acid lip augmentation, with emphasis on biomaterial behavior, anatomical precision, safety, reversibility, and documentation quality. ¹-¹⁴
Data sources: A targeted bibliographic corpus was assembled for interpretive synthesis. Priority was given to review articles, a systematic review and meta-analysis, lip-specific rheology and biomaterial characterization studies, imaging- and ultrasound-oriented follow-up papers, hyaluronidase literature, and anatomical references relevant to lip planning. ¹-¹⁴
Eligibility criteria: Sources were retained when they offered direct lip-specific applicability or when they clarified structure-property-performance relationships, safety, reversibility, or follow-up methodology with clear translational relevance to the lip context.
Methods of synthesis: This narrative review was organized around biomaterial identity, dynamic tissue behavior, vascular safety, and reversibility planning. A de-identified illustrative case was retained only to contextualize the framework. Serial standardized photographs obtained at baseline and at 15, 60, and 90 days were reviewed as longitudinal documentation; however, no instrument-based quantitative dataset was available, and the case is therefore presented as illustrative and non-probative.
Main findings: Predictable lip outcomes appear to depend less on injected volume alone than on the fit among rheological profile, network architecture, tissue dynamics, anatomical precision, and preparedness for complication management. Standardized photography remains useful for serial visual documentation, but volumetric claims require depth-resolved three-dimensional assessment, and ultrasound may strengthen both anatomical safety and post-procedural verification. In the illustrative case, standardized serial photographs suggest sustained improvement in contour definition, projection, and upper-to-lower lip proportion from 15 to 90 days after conservative subunit-based treatment with 1.0 mL of crosslinked hyaluronic acid. Because follow-up remained photographic rather than instrument-based, the case does not validate the higher evidentiary standard advocated by the framework.
Conclusion: Current evidence supports a conservative, anatomy-aware, biomaterial-centered approach to lip augmentation. Progress in the field depends on stronger comparative studies and more standardized reporting that better aligns biomaterial science, anatomical precision, reversibility planning, and documentation methodology.
Editorial audio summary
This audio summary is provided as complementary editorial content and does not replace the full peer-reviewed article.
Introduction
Lip augmentation occupies a distinctive place in orofacial practice because it involves aesthetics, oral competence, speech-related function, and fine contour perception within a highly mobile anatomical unit. The widespread use of crosslinked hyaluronic acid has sometimes encouraged a simplified volumization narrative; however, the lips are not a static compartment. They are subject to constant deformation, muscular recruitment, and high aesthetic scrutiny. Accordingly, a filler cannot be judged by volume alone or by generic descriptors such as soft or firm. ¹,²
Recent evidence reinforces this point. A systematic review and meta-analysis confirmed overall efficacy of hyaluronic acid lip fillers, but also documented frequent low-grade adverse events, including edema, firmness, tenderness, and bruising. ³ In this region, effectiveness and safety are inseparable, because modest variations in tissue plane, pressure, aliquot size, and recognition of vascular compromise may materially affect outcome quality.
The lips should therefore be approached as a dynamic biomechanical environment. Imaging-based follow-up has shown progressive changes in volume and projection after injection rather than a simple binary pattern of persistence versus disappearance. ⁴ Review-level discussion of in vivo post-injection change likewise suggests that tissue integration, redistribution, partial degradation, and local biological response all contribute to the observed clinical endpoint. ⁵
Material science and anatomy have thus become central to clinical interpretation. Lip-specific rheology, network organization, crosslinker chemistry, degradability to hyaluronidase, and anatomical variation in the labial arteries all influence how safely and naturally a filler can perform. ²,⁶-¹⁴ A clinically useful framework for lip augmentation should therefore integrate structure-property-performance logic, tissue mechanics, anatomical precision, reversibility planning, and procedural strategy rather than remain tied to a volume-centered model.
Objective and scope
This review starts from the premise that lip augmentation should not be discussed as product choice, anatomy, and technique in isolation. In clinical practice, performance is co-determined by biomaterial design, the mechanical demands of a highly mobile tissue bed, and local vascular risk.
The objective of this manuscript is to synthesize selected literature into a structured clinical framework for crosslinked hyaluronic acid lip augmentation, with emphasis on biomaterial behavior, anatomical precision, safety, reversibility, and translational relevance. A de-identified illustrative case is retained only as a secondary clinical example to contextualize the discussion; it is not presented as corroborative or confirmatory evidence. ¹,²,⁴,⁵,¹²-¹⁴
Methodological positioning and evidence prioritization
This manuscript is a narrative review based on a targeted interpretive synthesis of literature relevant to crosslinked hyaluronic acid lip augmentation. The source base was assembled from bibliographic material selected for conceptual and translational relevance rather than exhaustive coverage. The review privileged five source groups: (i) review articles and meta-analytic literature on lip fillers; (ii) lip-specific rheology and biomaterial characterization studies; (iii) imaging and ultrasound papers relevant to lip follow-up or safety; (iv) hyaluronidase and reversibility literature; and (v) anatomical or aesthetic references informing subunit-based planning. ¹-¹⁷
Sources were prioritized when they offered direct lip-specific applicability, clarified structure-property-performance relationships, strengthened safety or reversibility reasoning, or improved follow-up interpretation. Non-lip-specific filler literature was retained only when it contributed mechanistic or safety information with clear relevance to the lip context.
Because the purpose of the manuscript is conceptual integration rather than exhaustive evidence mapping, no formal systematic-search protocol or risk-of-bias assessment was undertaken. The evidentiary weight of each source category is therefore not treated as equivalent. Analytical, physicochemical, and in vitro studies are used to refine clinical reasoning, not to establish clinical superiority or deterministic performance in the lip context.
The illustrative case was retained because it provides a concrete, de-identified example of subunit-based planning, instrument selection, and longitudinal photographic follow-up. Its function is demonstrative only. It should be read as an application example of the proposed logic, not as validation of the framework or as proof of treatment effectiveness.
Structured synthesis of the literature
Biomaterial identity, rheology, and product-context fit
The literature increasingly supports the view that crosslinked hyaluronic acid lip filling should be treated as biomaterial implantation rather than simple soft-tissue enlargement. In a mobile anatomical site such as the lips, the clinically relevant question is not whether a filler can create volume in the abstract, but whether its material behavior is compatible with repetitive deformation, delicate contour demands, and the need to avoid rigidity or overprojection. ¹,²,⁵
Lip-specific rheological data are important because they show that products marketed for the same indication should not be presumed clinically equivalent by default. Differences in storage modulus, loss modulus, complex modulus, loss tangent, and cohesivity imply potential differences in support, spread, shape retention, and adaptation to motion. ² These variables should be interpreted as part of the material identity of the gel rather than as secondary technical details subordinate to injector preference.
That identity also depends on crosslinking architecture. Structural analysis has shown that degree of modification, crosslinker ratio, and network organization can be analytically characterized, enabling a more disciplined relationship between biomaterial engineering and clinical reasoning. ¹⁰ Experimental comparison of BDDE-crosslinked and PEGDE-crosslinked formulations further suggests that engineering choices may affect not only viscoelastic behavior but also biological response profiles. ¹¹ These findings help refine product-context reasoning, but they should not be overtranslated into direct clinical hierarchy claims for the lip context.
Material selection in lip augmentation is therefore best framed as a compatibility question: which rheological and structural profile appears most coherent for a given lip morphology, movement pattern, contour objective, and safety margin? ²,¹⁰,¹¹
Dynamic tissue behavior and the limits of follow-up claims
The dynamic nature of the lips complicates simplistic statements about longevity. Three-dimensional analysis coupled with sonography has demonstrated a progressive pattern of change in volume and projection after injection, which is more informative than a fixed statement about how long a product lasts. ⁴ Review literature on in vivo post-injection change likewise indicates that tissue integration, redistribution, partial degradation, and local biological response all contribute to the final clinical appearance. ⁵
These findings have methodological consequences. A field that aims for reproducibility should not rely exclusively on before-and-after photography or on subjective descriptors such as naturalness, softness, or persistence. Standardized photography remains useful because it permits serial visual documentation of contour, vermilion show, and symmetry when lighting, distance, expression, and head position are controlled. Yet photographic series remain inherently limited: they document projected appearance, not true three-dimensional tissue change.
Volumetric assessment requires depth-resolved three-dimensional acquisition rather than single-plane visual comparison. ⁴ Ultrasound may also strengthen documentation by identifying deposit location, vascular relationships, and, in selected cases, post-procedural filler topography. ⁶,¹³,¹⁴ Landmark-based frontal photography can improve two-dimensional reproducibility by supporting linear distances, proportional relationships, and symmetry analysis; however, it does not constitute volumetry and should not be interpreted as three-dimensional quantification.
For that reason, follow-up methodology should be described with terminological precision. A case may be documented by standardized photography alone, by photography plus two-dimensional morphometry, by three-dimensional surface capture, by ultrasound-based mapping, or by a multimodal combination. Conflating those levels of evidence weakens comparability and invites overinterpretation.
Vascular safety, plane control, and reversibility
Vascular safety remains the non-negotiable axis of lip augmentation. The superior and inferior labial arteries display variable depth, branching, and anastomotic patterns, which means that no injection plan should be assumed universally safe on the basis of instrument choice alone. ¹²,¹³ Cannulas may reduce the number of skin perforations and may favor broader product distribution, but they do not abolish risk. Needles may improve focal contour precision, but they demand heightened attention to plane, pressure, and aliquot size. ¹,³,⁶,⁷,¹²-¹⁴
This issue becomes especially important near the vermilion border. The region is aesthetically important but anatomically unforgiving, because small deviations in depth may move the operator from a superficial contouring intention toward a plane in which arterial structures may be encountered. The distinction between mucosal and more superficial subcutaneous intentions therefore matters clinically, even though those planes may not be consistently separated across individuals.
Contour work along the vermilion border should therefore be approached with conservative aliquots, low-pressure progression, continuous visual monitoring, and readiness to stop immediately if pain, blanching, or perfusion asymmetry becomes disproportionate. ¹,³,⁷,¹²-¹⁴ Ultrasound does not eliminate risk, but it can strengthen procedural accountability by improving knowledge of local vascular anatomy, helping localize filler deposits, and supporting management when complications occur. ⁶,¹³,¹⁴
Reversibility is not ancillary; it is part of pre-procedural planning. In vitro studies demonstrate that commercial fillers differ in hyaluronidase-mediated degradation kinetics, with implications for complication preparedness and management of overcorrection, asymmetry, or vascular events. ⁸,⁹ Review literature on hyaluronidase likewise underscores that reversal strategy should be incorporated into the initial treatment logic rather than reserved as an afterthought. ⁷
Illustrative case in clinical context
The de-identified illustrative case involved an 18-year-old woman seeking correction of thin lips and reduced vermilion show. Pre-procedure assessment demonstrated limited support and contour, and treatment was planned conservatively by anatomical subunits rather than indiscriminate global volumization. The selected product was 1.0 mL of crosslinked hyaluronic acid (Rennova Lift Ultra; Croma-Pharma), with the operative rationale centered on support, contour definition, and maintenance of natural proportion. ¹⁵-¹⁷
After antisepsis and bilateral infraorbital and mental blocks with 2% lidocaine plus vasoconstrictor, upper- and lower-lip subunits were approached with a combined cannula-needle strategy. A 22G x 50 mm microcannula was used for subcutaneous retrograde linear deposition in the upper lip and for controlled lower-lip refinement, whereas a 27G x 13 mm needle was reserved for more superficial refinement of the vermilion border, Cupid's bow, and lower outline. The total injected volume was 1.0 mL. The operative plan and volume allocation are summarized in Table 1.
Serial standardized photographs obtained at baseline and at 15, 60, and 90 days document the clinical evolution of the case across early and intermediate follow-up. Visually, the sequence suggests sustained improvement in contour definition, projection, and upper-to-lower lip proportion beyond the immediate post-procedural period, with preservation of a conservative and anatomically coherent result on serial photographic assessment.
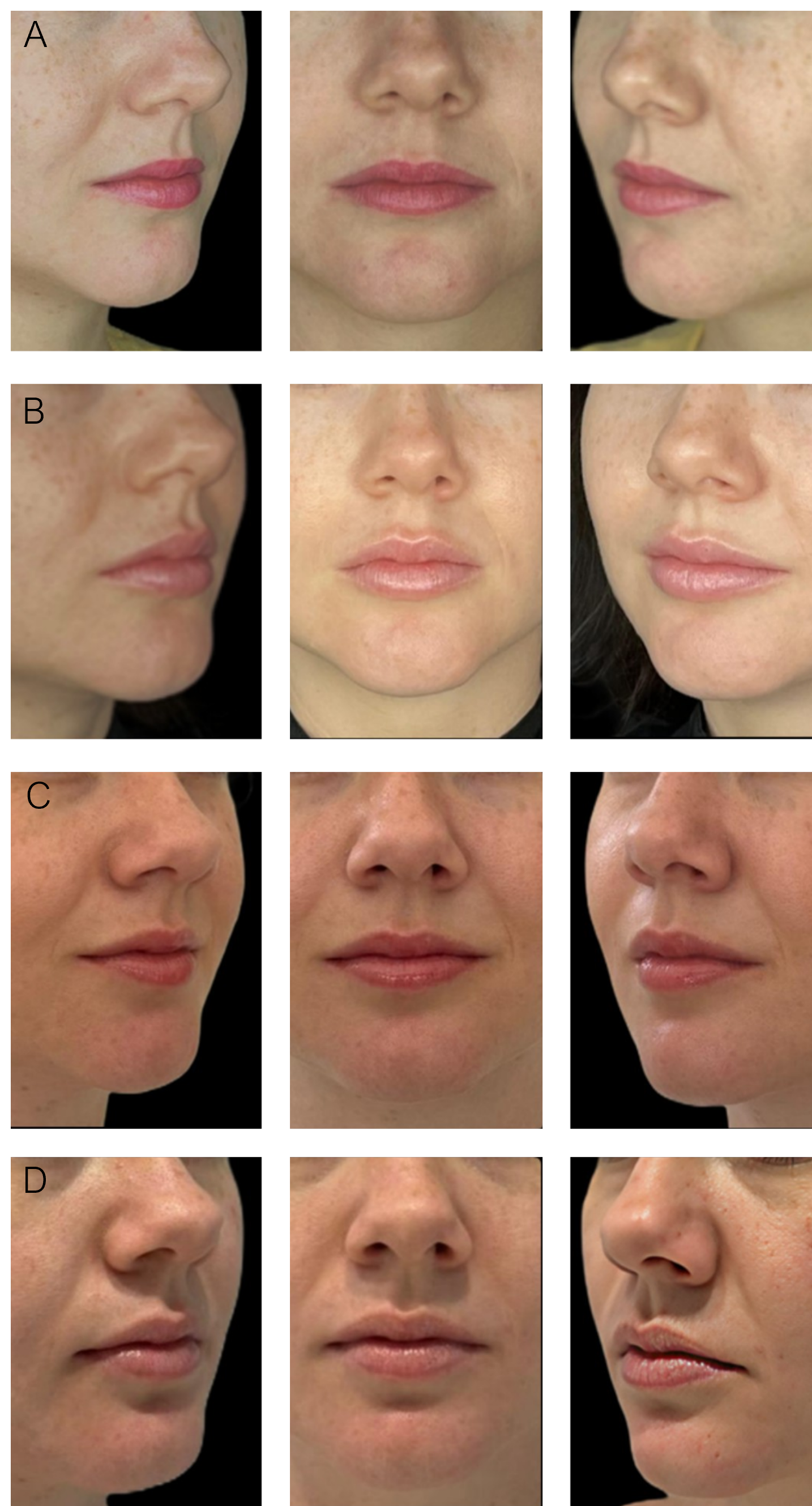
The case must nevertheless remain narrowly interpreted. It does not include three-dimensional volumetry, ultrasound-based assessment, or other instrument-based quantitative documentation. It therefore does not validate the proposed framework, does not establish product or technique superiority, and does not support quantitative claims about durability. Its value is demonstrative: it shows how an anatomy-aware, conservative, subunit-based logic may be applied in practice while making visible the documentation gap that still separates routine photographic follow-up from stronger instrumental confirmation.
| Region/objective | Instrument | Plane | Technique and mL |
|---|---|---|---|
| Upper lip - projection and volume | 22G x 50 mm microcannula | Subcutaneous | Two retrograde linear passes per side; 0.4 mL |
| Lower lip - refinement and tubercles | 22G x 50 mm microcannula | Subcutaneous | Retrograde linear pass plus mini-bolus support of the tubercles; 0.3 mL |
| Contour refinement | 27G x 13 mm needle | More superficial | Refinement of the vermilion border, Cupid's bow, and lower contour; 0.3 mL |
Critical appraisal and translational implications
The evidence base has matured in clinically meaningful ways, with more lip-specific rheology, imaging-based follow-up, ultrasound-informed safety discussion, hyaluronidase literature, and explicit biomaterial characterization than was typical in earlier filler publications. ²,⁴,⁶,⁷,¹⁰-¹⁴ This progression matters because it moves the field away from impressionistic description and toward properties that can be measured, audited, and interpreted.
Even so, the literature remains uneven. Products with distinct engineering profiles are often grouped together under broad labels; outcomes remain heterogeneous; follow-up is inconsistently standardized; and technique is still too often discussed as if it were separable from the material being injected. Lip-specific evidence remains thinner than the broader filler literature, which limits stronger conclusions about product-context fit. ¹-⁵,⁸-¹⁴
These weaknesses justify a restrained interpretation of efficacy. Acceptable short-term aesthetics and tolerability do not, by themselves, establish durable naturalness, mechanistic understanding, or robust safety generalizability. Higher-level clinical reasoning in lip augmentation depends on linking what was injected, where it was placed, how it behaves under movement, how it could be reversed, and how the outcome was documented.
A central translational implication of current evidence is that conservative lip augmentation is better conceptualized as a biomaterial deployment problem than as volume delivery alone. Product selection should therefore be justified against tissue mobility, subunit-specific support requirements, reversibility strategy, and vascular risk. Standardized photography remains necessary, but stronger reporting should also specify the evidentiary level of follow-up: photographic only, photography plus two-dimensional morphometry, three-dimensional surface capture, ultrasound-based mapping, or a multimodal combination.
To support more consistent reporting, Table 2 summarizes minimum documentation elements that would strengthen interpretability without overstating the current evidence base.
| Domain | Minimum element to report | Why it matters |
|---|---|---|
| Product identity | Commercial product and manufacturer | Prevents vague grouping of non-equivalent materials |
| Biomaterial class | Crosslinked hyaluronic acid formulation | Clarifies the implant category under discussion |
| Rheological rationale | Reason for choosing the gel profile for lip use | Links material choice to the clinical objective |
| Structure-property rationale | Cohesivity and crosslinking logic, when available | Connects engineering to expected in vivo behavior |
| Anatomical target | Subunit or contour objective | Prevents indiscriminate global volumization language |
| Injection plane/depth intention | Intended plane for each procedural step | Central to safety and performance interpretation |
| Instrument used | Needle or cannula specification | Required for technical reproducibility |
| Volume allocation | Total volume and regional distribution | Allows interpretation beyond total mL alone |
| Safety strategy | Monitoring approach and stop criteria | Anchors risk management |
| Reversibility preparedness | Availability and planning for hyaluronidase | Makes complication response explicit |
| Documentation method | Standardized photography; two-dimensional morphometry when applicable; three-dimensional capture and/or ultrasound when available | Clarifies the evidentiary level of follow-up |
| Follow-up intervals | Time points reported | Improves interpretation of persistence |
| Complication monitoring | Immediate and delayed surveillance elements | Essential for safety reporting |
| Reported outcomes | Contour, projection, proportion, symmetry, tolerance, and any objective metric used | Encourages consistent endpoint reporting |
Limitations
This manuscript is limited by its narrative design, selective evidentiary base, and lack of a formal systematic-search protocol or risk-of-bias assessment. The underlying literature is also heterogeneous with respect to product engineering, follow-up standardization, and lip-specific outcome reporting.
The illustrative case adds clinical context and includes standardized serial photographic follow-up through 90 days. This strengthens longitudinal documentation when compared with a single short-term photographic checkpoint. However, the case remains a single documentation-based example and does not include three-dimensional volumetry, ultrasound-based assessment, or other instrument-based quantitative validation.
These constraints materially restrict comparative, predictive, and durability-oriented conclusions. Accordingly, the manuscript cannot support product ranking, strong comparative claims between techniques, or long-term mechanistic conclusions about persistence.
Conclusion
Crosslinked hyaluronic acid lip augmentation is best understood as biomaterial implantation in a highly mobile and vascularly sensitive anatomical site. Clinically credible outcomes arise less from injected volume alone than from the fit among gel properties, tissue mechanics, anatomical precision, conservative execution, readiness for reversal, and documentation quality.
A more mature clinical literature will depend on clearer integration of biomaterial science, lip-specific anatomy, reversibility planning, and appropriately stratified follow-up methodology. The available evidence supports a conservative, anatomy-aware, biomaterial-centered approach while underscoring the need for stronger comparative studies and more standardized outcome reporting. In the present manuscript, serial standardized photographs through 90 days strengthen the illustrative case as longitudinal documentation, but they do not convert it into instrumentally confirmatory evidence.
Declarations
Ethics approval: Not applicable to this narrative review. The illustrative case was included with publication authorization and without direct identifiers.
Consent to participate: Written informed consent was obtained from the patient for clinical documentation and scholarly use of de-identified material.
Consent for publication: Written informed consent authorizing journal publication of the de-identified clinical images included in Figure 1 was obtained from the patient and is held on file; it can be provided to the editor on request.
Conflicts of interest: The author declares no conflicts of interest relevant to this narrative review.
Funding: No external funding.
Data availability: All data supporting the conclusions of this manuscript are contained within the article. Additional de-identified clinical material is not publicly available because of privacy considerations.
Code availability: Not applicable.
AI use disclosure: AI-assisted language editing was used during drafting and revision of the manuscript. Standardized photographic workflow procedures were used for session traceability, metadata organization, and landmark-compatible two-dimensional image annotation in the illustrative case. The authors reviewed and verified the final manuscript and take full responsibility for all content, citations, interpretations, figure descriptions, and conclusions.
Author contributions: Kamila Salomão Galdino conceived the review, conducted the literature synthesis, interpreted the evidence, prepared the manuscript, and approved the final version.
Acknowledgements: None.
References
- Cooper H, Gray T, Fronek L, Witfill K. Lip augmentation with hyaluronic acid fillers: a review of considerations and techniques. J Drugs Dermatol. 2023;22(1):23-29. doi:10.36849/JDD.6304.
- Nogueira PMS, de Lima Romeiro R, Cortelli SC. Rheological properties of hyaluronic acid fillers for lip volumization. Aesthet Plast Surg. 2025;49(21):5877-5883. doi:10.1007/s00266-025-05121-2.
- Wen YE, Perez Rivera LR, Wyatt HP, Lee WY, Oh C, Boyd CJ, et al. Efficacy and safety of hyaluronic acid lip fillers: a systematic review and meta-analysis of randomized controlled trials. Aesthet Surg J. 2025:sjaf224. doi:10.1093/asj/sjaf224.
- Kim JS. Changes in volume of lips in 3-dimensional analysis and projection of lips in sonography after injection of particle-type hyaluronic acid filler utilizing a 9-point injection technique. Aesthet Surg J Open Forum. 2024;6:ojae076. doi:10.1093/asjof/ojae076.
- Hong GW, Wan J, Chang K, Park Y, Yi KH. Decomposition and changes in in vivo post-HA filler injection: a review. J Cosmet Dermatol. 2025;24(1):e16652. doi:10.1111/jocd.16652.
- Cavallieri FA, Balassiano LKA, Munhoz G, Tembra MF, Wortsman X. Ultrasound in aesthetics: filler and non-filler applications. Semin Ultrasound CT MR. 2024;45(3):251-263. doi:10.1053/j.sult.2023.11.005.
- Kroumpouzos G, Treacy P. Hyaluronidase for dermal filler complications: review of applications and dosage recommendations. JMIR Dermatol. 2024;7:e50403. doi:10.2196/50403.
- Demir A, Tayfun V. In vitro analysis of hyaluronidase degradation of commercial hyaluronic acid fillers: considerations in lip augmentation. J Craniofac Surg. 2025 Aug 20. doi:10.1097/SCS.0000000000011837.
- Faivre J, Wu K, Gallet M, Sparrow J, Bourdon F, Gallagher CJ. Comparison of hyaluronidase-mediated degradation kinetics of commercially available hyaluronic acid fillers in vitro. Aesthet Surg J. 2024;44(6):NP402-NP410. doi:10.1093/asj/sjae032.
- Lee W, Yang EJ. Structural analysis of hyaluronic acid fillers using nuclear magnetic resonance: implications for quality control and clinical performance. Polymers (Basel). 2024;16(20):2878. doi:10.3390/polym16202878.
- Kim DH, Jeong CH, Han JH, Lim SJ, Kwon HC, Kim YJ, et al. Comparative toxicity study of hyaluronic acid fillers crosslinked with 1,4-butanediol diglycidyl ether or poly(ethylene glycol) diglycidyl ether. Int J Biol Macromol. 2025;296:139620. doi:10.1016/j.ijbiomac.2025.139620.
- Cotofana S, Pretterklieber B, Lucius R, Frank K, Haas M, Schenck TL, et al. Distribution pattern of the superior and inferior labial arteries: impact for safe upper and lower lip augmentation procedures. Plast Reconstr Surg. 2017;139(5):1075-1082. doi:10.1097/PRS.0000000000003244.
- Wu WT, Chang KV, Nanka O, Chang HC, Ricci V, Mezian K, et al. Lip sonoanatomy and relevance to aesthetic filler injections: a pictorial review. J Cosmet Dermatol. 2025;24(4):e70164. doi:10.1111/jocd.70164.
- Almushayt SJ. The role of ultrasound in facial hyaluronic acid dermal filler injections - a review article. Clin Imaging. 2025;119:110413. doi:10.1016/j.clinimag.2025.110413.
- Jacono AA. A new classification of lip zones to customize injectable lip augmentation. Arch Facial Plast Surg. 2008;10(1):25-29. doi:10.1001/archfaci.10.1.25.
- Popenko NA, Tripathi PB, Devcic Z, Karimi K, Osann K, Wong BJF. A quantitative approach to determining the ideal female lip aesthetic and its effect on facial attractiveness. JAMA Facial Plast Surg. 2017;19(4):261-267. doi:10.1001/jamafacial.2016.2049.
- Kim SW, Rousso JJ. Determining the 2-dimensional threshold for perception of artificial-appearing lips. JAMA Facial Plast Surg. 2017;19(5):392-398. doi:10.1001/jamafacial.2017.0052.