Abstract
Objective: To critically reassess the use of botulinum toxin type A (BoNT/A) for gummy smile, with emphasis on anatomical targeting, protocol variability, and the conditions required for reproducible outcome measurement.
Data sources: Literature addressing etiologic phenotyping of gummy smile, elevator-muscle anatomy, BoNT/A formulation and preparation, targeted clinical application, and digital approaches to image-based outcome capture.
Eligibility criteria: Studies and technical sources were prioritized when they directly informed five linked domains: phenotype selection, anatomical rationale, product handling, delivery variables, and the reliability of endpoint documentation.
Methods of synthesis: Critical narrative review integrating anatomical, clinical, pharmaceutical, and measurement-oriented evidence relevant to the translational interpretation of BoNT/A therapy for gummy smile.
Main findings: The literature supports temporary gingival-display reduction in selected muscular cases and offers a credible anatomical rationale for targeting the Yonsei region. However, universality of the point is not supported. Morphometric variation, heterogeneous smile-task architecture, non-equivalent product preparation, and incompletely reported dose-volume-depth relationships remain major interpretive barriers.
Conclusion: BoNT/A remains a defensible minimally invasive option for compatible muscular etiologies, but the field will mature only when anatomical targeting, product handling, dose-volume-depth architecture, smile-task definition, and digitally auditable outcome measurement are integrated within one reproducible protocol.
Keywords
Keywordsbotulinum toxin type A; gummy smile; Yonsei point; digital workflow; facial imaging; outcome measurement.
Audio summary
This audio summary is provided as complementary editorial content and does not replace the full peer-reviewed article.
Read audio transcript
Welcome to the Journal of Digital Health and Advanced Biomaterials audio series.
In this episode, we present Diego Silva’s article on botulinum toxin type A for gummy smile, with emphasis on anatomical targeting, protocol variability, and reproducible outcome measurement.
The article critically reassesses the use of botulinum toxin type A in selected muscular cases of excessive gingival display. It does not treat gummy smile as a single diagnosis, but as a clinical presentation that may result from different anatomical and functional conditions.
One of the central messages is that patient selection matters. Botulinum toxin can be a defensible minimally invasive option when the main cause is muscular hyperactivity, but it should not be interpreted as an appropriate solution for every form of gingival display.
The review also examines the Yonsei point as an important anatomical reference. This point provides a useful clinical shorthand because it corresponds to the functional convergence of upper-lip elevator muscles. However, the article cautions that the Yonsei point should not be treated as a universal or fixed coordinate.
Anatomical variability, smile phenotype, injection depth, dose, volume, product handling, and follow-up documentation all influence clinical interpretation. For that reason, the paper argues that an apparently simple injection technique actually depends on a complex therapeutic system.
The article also discusses the molecular and pharmacological basis of botulinum toxin type A. Its clinical effect depends on temporary chemodenervation through inhibition of acetylcholine release, but the toxin is not used in practice as an abstract molecule. It is delivered as a formulated, reconstituted, and injected product, which makes preparation and delivery variables important for reproducibility.
A major contribution of the review is its focus on outcome measurement. The literature often reports clinical improvement, but photographic protocols, smile tasks, follow-up intervals, calibration methods, and repeatability procedures remain heterogeneous. These differences can make results difficult to compare across studies.
The review therefore proposes a more disciplined reporting framework. Future studies should describe phenotype selection, anatomical targeting logic, product formulation, reconstitution, injected units, volume, depth, smile-task architecture, image-capture protocol, measurement method, repeatability, follow-up, and safety events.
The conclusion is clear. Botulinum toxin type A remains a plausible and useful option for compatible muscular etiologies of gummy smile, but the field will advance only when anatomical reasoning, product handling, dose-volume-depth architecture, and digitally auditable outcome measurement are integrated into reproducible clinical protocols.
Rather than multiplying named injection points, this article calls for better documentation before, during, and after treatment. Its central contribution is to move the discussion from isolated technique toward anatomical precision, methodological transparency, and reproducible evidence.
This was the audio summary of Diego Silva’s article, published in the Journal of Digital Health and Advanced Biomaterials. Thank you for listening.
Introduction
Among minimally invasive options for gummy smile, botulinum toxin type A (BoNT/A) is attractive because it is reversible, rapid, and often effective in appropriately selected muscular phenotypes. Yet the apparent simplicity of the injection obscures the complexity of the therapeutic system: etiologic selection, landmark choice, formulation, dilution, injected volume, depth, symmetry, and image capture all influence the clinical results.5-13,20,23
That complexity is especially consequential in the perioral region. The therapeutic window is narrow, esthetic penalties are immediate, and small technical differences can be mistaken for biologic inconsistency. The result is a literature in which the treatment often seems more reproducible than the endpoint used to judge it.10-13,23,27
This review therefore addresses a more stringent question: not simply whether BoNT/A may reduce gingival exposure in selected muscular cases, but which variables determine whether the Yonsei point functions as a reproducible clinical target or remains an anatomical approximation with limited transferability.14-19,22,26,27
Review objective
This review critically reassesses BoNT/A for gummy smile from a translational standpoint. Rather than treating the Yonsei point as an isolated technical maneuver, it examines the interaction among phenotype, anatomy, pharmaceutical handling, and endpoint capture, asking where the current literature is genuinely robust and where it remains methodologically fragile.10-13,20,23,27
Review method and interpretive strategy
This manuscript was prepared as a critical narrative review rather than a systematic review or quantitative synthesis. The literature was selected for direct relevance to five linked domains: etiological phenotyping of gummy smile, upper-lip elevator anatomy, BoNT/A formulation and preparation, clinical performance of targeted injection strategies, and the reliability of imaging-based outcome capture.
Evidence was not weighted only by reported clinical effects. Additional interpretive value was assigned to studies that clarified how image acquisition, calibration, metadata structure, or computational post-processing affected endpoint trustworthiness.10-13,23,27
Thematic synthesis of the literature
Gummy smile is a clinical presentation, not a unitary diagnosis. Excessive gingival display may reflect vertical maxillary excess, dentoalveolar disproportion, altered passive eruption, lip hypermobility, or hyperactivity of the upper-lip elevator muscles. This distinction is not taxonomic only. It determines whether BoNT/A is mechanistically relevant and therefore whether clinical response can be interpreted as treatment success rather than diagnostic mismatch.5-7,10-13
Polo’s studies were important not only because they documented reduction in gingival display, but because they made patient selection central to the interpretation of response. Later prospective studies, controlled designs, and pooled analyses support short-term benefit in selected muscular cases, but they do not erase heterogeneity in indication, technique, or outcome capture.5-13
Anatomical basis of the Yonsei point
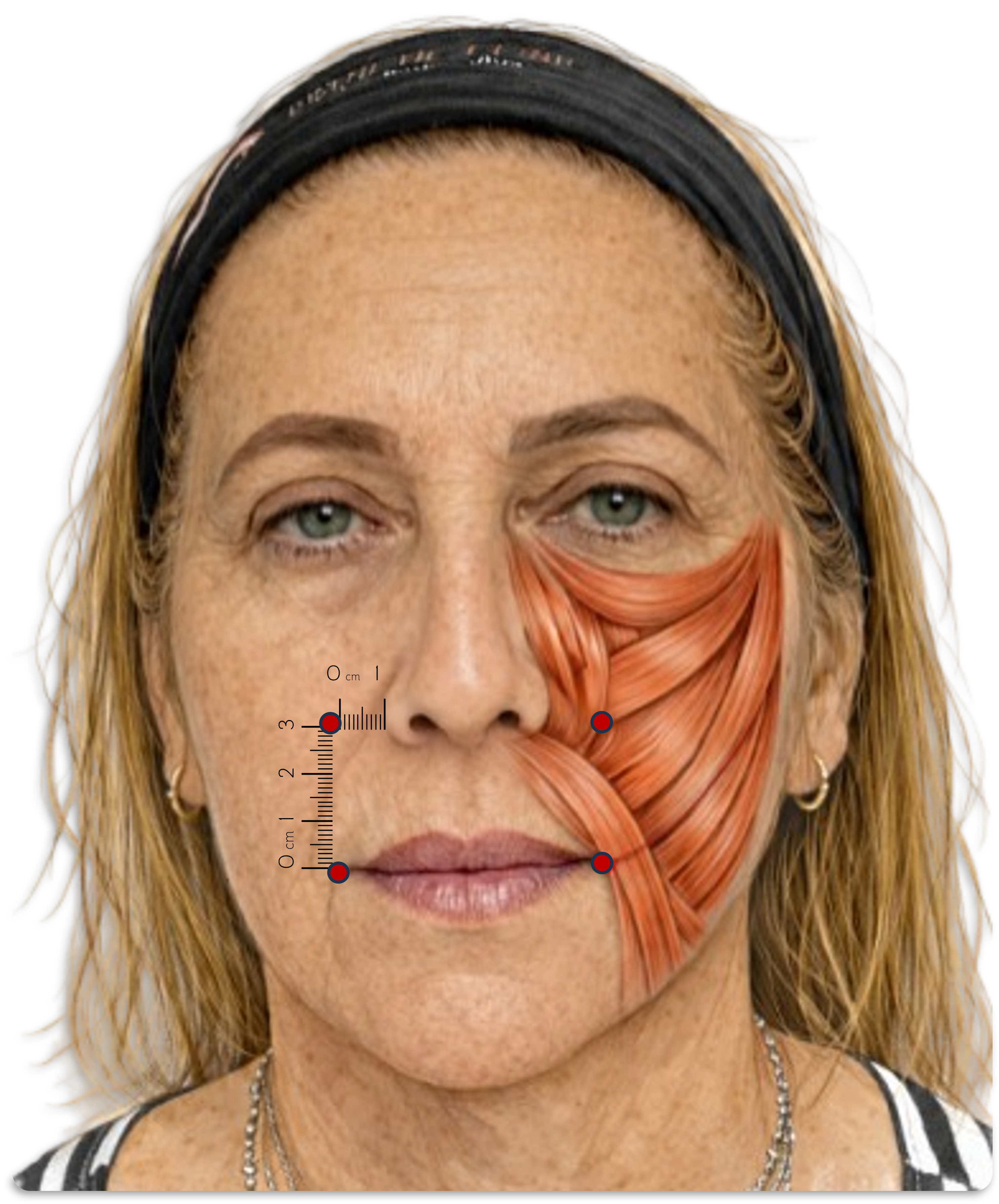
The Yonsei point was a major attempt to subject perioral injection to anatomical reasoning. In the foundational study, Hwang and colleagues mapped the levator labii superioris alaeque nasi, levator labii superioris, and zygomaticus minor, and proposed a single surface target corresponding to their zone of functional convergence. Its procedural simplicity, however, should not be mistaken for universality.14-17
Recently controlled comparison and expert consensus preserve the lateral alar targeting logic, but they also show that injection plane, per-point volume, and measurement workflow must be treated as protocol-defining variables rather than as technical afterthoughts.19,26,27
Molecular and pharmacological basis of BoNT/A
Any clinical account of BoNT/A must begin with its modular protein architecture. Heavy-chain and light-chain domains mediate neuronal binding, internalization, translocation, and catalytic activity; after entry into cholinergic terminals, the light chain cleaves SNAP-25, blocks acetylcholine release, and produces temporary chemodenervation.1,2
This mechanistic basis matters because the toxin never reaches practice as an abstract molecule. It is used as a formulated, lyophilized, reconstituted, and injected product. Preparation conditions and physicochemical context may therefore contribute to variable clinical expression and should not be treated as negligible background variables.3,4,20,21
Clinical evidence and critical interpretation
The clinical literature supports, with moderate consistency, that BoNT/A can temporarily reduce gingival exposure in patients with a predominantly muscular component. What remains unresolved is not whether an effect can occur, but why effect size, symmetry, durability, and esthetic naturalness vary so widely across reports and patients.6-13
The translational problem is therefore not the absence of an anatomical hypothesis, but the persistent mismatch between anatomical plausibility and measurement rigor. Read together, contemporary studies support the Yonsei point as a useful anatomical shorthand, not as a definitive or universally transferable coordinate.14-19,22
Key studies
Table 1. Key studies informing the anatomical rationale, clinical performance, measurement validity, and workflow traceability relevant to Yonsei-point use in gummy-smile management.
| Study | Design | Main contribution | Critical relevance to the present manuscript |
|---|---|---|---|
| Hwang et al., 2009 | Anatomical study | Mapped LLSAN, LLS, and ZMi and proposed the Yonsei point as an area of functional convergence. | Foundational anatomical rationale supports the logic of precision, but does not authorize automatic universalization of the point. |
| Polo, 2008 | Prospective study | Documented neuromuscular correction of gummy smile with marked short-term reduction in gingival exposure. | Key evidence for transient efficacy, but not sufficient to resolve anatomical or methodological comparability across techniques. |
| Mazzuco and Hexsel, 2010 | Prospective study | Linked gingival exposure pattern to the muscular territory involved. | Important for shifting discussion from an isolated point to functional phenotyping of the smile pattern. |
| Al Wayli, 2019 | Clinical study | Applied a single Yonsei-point strategy with favorable outcomes in selected muscular phenotypes. | Useful for clinical dissemination, but interpretation remains dependent on etiologic selection and photographic standardization. |
| Booysen et al., 2023 | Population morphometric study | Tested standardization of the point in a White South African population and demonstrated topographic variability. | One of the strongest warnings against dogmatic use of the Yonsei point as a universal landmark. |
| Gong et al., 2024 | Randomized controlled trial | Compared Yonsei-point injection with classical LLSAN targeting in a longitudinal design. | Signals movement toward more controlled anatomical comparison, but persistent variability in technique and measurement remains unresolved. |
| Ayaz et al., 2020 | Measurement-reliability study | Compared 2D photography with 3D soft-tissue imaging and showed that protocolized 2D capture can be reliable, while 3D surface imaging generally provides lower error. | Supports the argument that endpoint technology is not neutral and that facial metrology should be treated as a core translational variable. |
| Goncalves et al., 2026 | Workflow/reporting study | Described a session-based web module for standardized clinical imaging and automated unit totalization in orofacial harmonization. | Illustrates how traceable digital documentation can bridge anatomical targeting and reproducible reporting in real-world injectable practice. |
| Myung et al., 2021 | Narrative review | Synthesized study-selection, smile-induction, measurement-site, and media heterogeneity across the gummy-smile BoNT literature. | Useful bridge between classical clinical reports and the present emphasis on standardized capture, scripted smile tasks, and reproducible measurement. |
| Rho et al., 2025 | Expert consensus guideline | Recommended small bilateral dosing near the lateral alar region and discussed patient selection, philtrum length, injection depth, and severity-based adaptation. | Provides contemporary protocol nuance and reinforces that point location, plane, and risk architecture must be specified rather than assumed. |
Abbreviations: LLSAN, levator labii superioris alaeque nasi; LLS, levator labii superioris; ZMi, zygomaticus minor.
Outcome measurement, digital validity, and translational priority
If one bottleneck runs through this literature, it is outcome measurement. Systematic reviews and meta-analyses repeatedly identify non-equivalent photographic intervals, inconsistent smile tasks, and sparse reporting of calibration or repeatability.10-13,23,27
From a digital-health perspective, the relevant shift is from informal landmark-based documentation to digitally auditable clinical targeting. Contemporary workflows can link standardized capture, structured metadata, session-based point mapping, landmark annotation, calibration fields, and exportable records within a single traceable object.14,24
Minimum reporting checklist
Table 2. Proposed minimum reporting checklist for BoNT/A studies addressing gummy smile.
| Domain | Minimum items to report | Why it matters |
|---|---|---|
| Phenotype and indication | Predominant etiology; inclusion criteria for muscular cases; whether vertical maxillary excess, altered passive eruption, or dentoalveolar factors were excluded or co-managed. | BoNT/A is mechanistically relevant only for compatible phenotypes; mixed etiologies distort apparent efficacy. |
| Anatomical targeting rationale | Point(s) selected; muscles intended to be modulated; side-to-side symmetry logic; whether the Yonsei point was used as fixed reference, adapted landmark, or comparator. | Named landmarks are not analytically interchangeable unless their anatomical logic is explicit. |
| Product and reconstitution | BoNT/A formulation; nominal vial size; reconstitution volume; resulting concentration; time from reconstitution to injection; relevant lot or traceability data when appropriate. | Preparation conditions affect delivered product behavior and are essential for cross-study comparability. |
| Injection map and dose logic | Units per side; volume per point; depth; needle or cannula details if relevant; number of points; whether low-dose-plus-retouch or fixed-dose strategy was used. | Dose alone does not describe delivery; volume, depth, and point architecture condition effect and diffusion behavior. |
| Capture protocol | Camera or focal-length equivalent; working distance; lighting and exposure conditions; background; head orientation; smile task; timepoints captured; file naming or session organization. | A weakly controlled facial endpoint can mimic or mask treatment effect. |
| Measurement protocol | Calibration method; software; landmarks or linear measurements used; whether analysis was 2D or 3D; operational definition of gingival display or lip-elevation endpoint. | Outcome definition determines whether the reported change is measurable, reproducible, and clinically interpretable. |
| Repeatability and quality control | Operator training; inter- or intra-operator agreement; intraclass correlation coefficient (ICC), technical error of measurement (TEM), or Bland-Altman analysis where applicable; post-registration error or QC rules for 3D pipelines; handling of unusable captures. | Measurement reproducibility validates the workflow itself and limits overinterpretation of visually persuasive but unstable endpoints. |
| Follow-up and safety | Exact follow-up intervals; retreatment policy; adverse events; asymmetry handling; whether photographs at later timepoints used the same capture rules. | Durability, symmetry, and safety cannot be interpreted when time-linked documentation is inconsistent. |
| Smile-task architecture | Exact expression state(s) requested; induction method; whether repeated capture or video-assisted frame selection was used for maximum smile; analytic frame-selection rule. | Non-equivalent smile tasks can create apparent treatment differences that are behavioral rather than pharmacologic. |
| Injection plane and needle geometry | Tissue plane (deep subcutaneous, superficial subdermal/intradermal, or intramuscular if justified); needle gauge/length; per-point volume and concentration. | Nominal units do not fully describe delivery architecture or diffusion risk in the perioral region. |
This checklist is intended to operate a small field without unnecessary complexity. Its purpose is to condense into a single reporting framework the variables that determine interpretability: phenotype, targeting logic, product handling, capture conditions, calibration, repeatability, follow-up, and safety.
Repeatability should be documented with intra- and/or inter-operator agreement metrics, such as the intraclass correlation coefficient (ICC) for repeated continuous measurements, with technical error of measurement (TEM) or Bland-Altman analysis used where appropriate.
Knowledge gaps and practical research agenda
The key unresolved question is no longer whether BoNT/A may work in gummy smile, but why it works unevenly across patients and study designs. Future work should prioritize anatomically explicit cohort selection, full reporting of product preparation and delivery variables, and prospectively defined endpoints with documented repeatability.
A practical agenda is clear: standardized frontal and profile capture; explicit smile-task instructions; internal metric calibration when justified; validated 3D surface imaging where available; session-based injection mapping; and prospective comparisons between surface-landmark targeting and digitally enhanced localization strategies.23,24
Conclusion
BoNT/A is a defensible minimally invasive option for gummy smile when confined to compatible muscular etiologies and interpreted through a disciplined, anatomically informed protocol. Current evidence supports anatomical plausibility and short-term clinical usefulness in selected cases, but it does not justify anatomical universalization or tacit equivalence among protocols that differ in phenotyping, product handling, delivery, and endpoint capture.
The field will advance most convincingly not by multiplying named target points, but by improving the precision of what is documented before, during, and after injection. The strongest next step is a framework that links phenotype classification, anatomically explicit targeting, full reporting of preparation and delivery variables, calibrated digital capture, structured metadata, and repeatable measurement within one auditable workflow.
Required statements
- Ethics approval
- Not applicable.
- Consent to participate
- Not applicable.
- Consent for publication
- Written authorization was obtained for the use of the de-identified clinical photographs presented in the illustrative figures.
- Competing interests
- The author declares no competing interests.
- Funding
- This review received no external funding.
- Data availability statement
- No new datasets were generated or analyzed for this review article.
- Code availability statement
- Not applicable.
- AI use disclosure
- Generative AI-assisted language editing was used during manuscript revision. The author reviewed, validated, and approved the final content and remains fully responsible for the manuscript.
References
References are set in Vancouver/NLM style and kept compact without breaking the manuscript sequence.
- Lacy DB, Tepp W, Cohen AC, DasGupta BR, Stevens RC. Crystal structure of botulinum neurotoxin type A and implications for toxicity. Nat Struct Biol. 1998;5(10):898-902. doi:10.1038/2338.
- Gregory KS, Acharya KR. A comprehensive structural analysis of Clostridium botulinum neurotoxin A cell-binding domain from different subtypes. Toxins (Basel). 2023;15(2):92. doi:10.3390/toxins15020092.
- Hada S, Lee JC, Lee EC, Ji S, Kim NA, Jeong SH, et al. Dissociation mechanics and stability of type A botulinum neurotoxin complex by means of biophysical evaluation. J Pharm Investig. 2022;52:453-63. doi:10.1007/s40005-022-00570-2.
- Ellis K, Thach T, Gallagher CJ. Biochemical stability and microbial control of reconstituted daxibotulinumtoxinA-lanm for injection. Toxins (Basel). 2023;15(12):683. doi:10.3390/toxins15120683.
- Polo M. Botulinum toxin type A in the treatment of excessive gingival display. Am J Orthod Dentofacial Orthop. 2005;127(2):214-8.
- Polo M. Botulinum toxin type A (Botox) for the neuromuscular correction of excessive gingival display on smiling (gummy smile). Am J Orthod Dentofacial Orthop. 2008;133(2):195-203.
- Mazzuco R, Hexsel D. Gummy smile and botulinum toxin: a new approach based on the gingival exposure area. J Am Acad Dermatol. 2010;63(6):1042-51. doi:10.1016/j.jaad.2009.09.043.
- Cengiz AF, Goymen M, Akcali C. Efficacy of botulinum toxin for treating a gummy smile. Am J Orthod Dentofacial Orthop. 2020;158(1):50-8. doi:10.1016/j.ajodo.2019.07.014.
- Costa AB, Romansina D, Ramalho J, Pereira P, Tedesco TK, Morimoto S, et al. Botulinum toxin A in the management of a gummy smile: a clinical controlled preliminary study. Aesthet Surg J. 2022;42(4):421-30. doi:10.1093/asj/sjab342.
- Chagas TF, Almeida NV, Lisboa CO, Ferreira DMTP, Mattos CT, Mucha JN. Duration of effectiveness of botulinum toxin type A in excessive gingival display: a systematic review and meta-analysis. Braz Oral Res. 2018;32:e30. doi:10.1590/1807-3107bor-2018.vol32.0030.
- Zengiski ACS, Basso IB, Cavalcante-Leao BL, Stechman-Neto J, Santos RS, Guariza-Filho O, et al. Effect and longevity of botulinum toxin in the treatment of gummy smile: a meta-analysis and meta-regression. Clin Oral Investig. 2022;26(1):109-17. doi:10.1007/s00784-021-04223-w.
- Rojo-Sanchis C, Montiel-Company JM, Tarazona-Alvarez B, Haas-Junior OL, Peiro-Guijarro MA, Paredes-Gallardo V, et al. Non-surgical management of the gingival smile with botulinum toxin A: a systematic review and meta-analysis. J Clin Med. 2023;12(4):1433. doi:10.3390/jcm12041433.
- Wang XY, Zou YR, Yuan MN, Huang HY, Han XF, Gong X. Dose and injection site of botulinum toxin type A for gummy smile management: a systematic review and bibliometric analysis. Toxicon. 2024;249:108058. doi:10.1016/j.toxicon.2024.108058.
- Hwang WS, Hur MS, Hu KS, Song WC, Koh KS, Baik HS, et al. Surface anatomy of the lip elevator muscles for the treatment of gummy smile using botulinum toxin. Angle Orthod. 2009;79(1):70-7. doi:10.2319/021208-70.1.
- Duruel O, Ataman-Duruel ET, Tozum TF, Berker E. Ideal dose and injection site for gummy smile treatment with botulinum toxin-A: a systematic review and introduction of case study. Int J Periodontics Restorative Dent. 2019;39(4):e167-73. doi:10.11607/prd.3918.
- Al Wayli H. Versatility of botulinum toxin at the Yonsei point for the treatment of gummy smile. Int J Esthet Dent. 2019;14(1):86-95.
- Gupta D, Kohli S. The Yonsei point: a precision approach to gummy smile correction with botulinum neurotoxin. Indian Dermatol Online J. 2019;10(5):560-2. doi:10.4103/idoj.IDOJ_29_19.
- Booysen M, Becker P, Lemmer J, Lemmer P. Establishing standards for Yonsei point in a White South African population. Clin Exp Dent Res. 2023;9(5):1216-24. doi:10.1002/cre2.790.
- Gong X, Tang HN, Zhang AR, Wang Z, Tang ZH, Han XF, et al. Application of botulinum toxin at the Yonsei point for the treatment of gummy smile: a randomized controlled trial. Plast Reconstr Surg. 2024;153(4):711e-21e. doi:10.1097/PRS.0000000000010623.
- Picelli A, Tamburin S, Di Censo R, Smania N, Filippetti M. Does the diffusion profile differ between botulinum toxin type A formulations? Implications for the management of post-stroke spasticity. Toxins (Basel). 2024;16(11):480. doi:10.3390/toxins16110480.
- Gardner A, Sneller M, Tepp WH, Barbieri JT, Pellett S. Botulinum neurotoxin Light Chain/A1 uses fast synaptic vesicle cycling to cleave plasma membrane-bound SNAP-25. Commun Biol. 2025;8:1383. doi:10.1038/s42003-025-08633-4.
- Wan J, Yoon SE, Cartier H, Garson S, Yi KH. The Yonsei point: a precision approach to gummy smile correction with botulinum neurotoxin. J Craniofac Surg. 2025 Oct;36(7):e852-5. doi:10.1097/SCS.0000000000011253.
- Ayaz I, Shaheen E, Aly M, Shujaat S, Gallo G, Coucke W, et al. Accuracy and reliability of 2-dimensional photography versus 3-dimensional soft tissue imaging. Imaging Sci Dent. 2020;50(1):15-22. doi:10.5624/isd.2020.50.1.15.
- Goncalves L, Silva D, Galdino K. Mapping botulinum toxin injection sites and automated unit totalization in orofacial harmonization: a session-based web module for standardized, traceable clinical imaging. Derecho y Cambio Social. 2026;23(88):1-19. doi:10.54899/dcs.v23i88.4842.
- Silva D, Goncalves L, Galdino KS. Gummy smile modulation with onabotulinumtoxinA at the Yonsei point after aesthetic ceramic rehabilitation: a case report. Derecho y Cambio Social. 2026;23(87):1-16. doi:10.54899/dcs.v23i87.4768.
- Rho NK, Bae GY, Choi MS, Chung WK, Kim HY, Kim HM, et al. Consensus on the cosmetic use of a novel botulinum neurotoxin type A product (NEWLUX®) for facial expression muscles: 2024 guidelines and discussions by Korean experts. Toxins (Basel). 2025;17(2):61. doi:10.3390/toxins17020061.
- Myung Y, Woo K, Kim ST. Treatment of gummy smile using botulinum toxin: a review. J Dent Rehabil Appl Sci. 2021;37(2):61-72. doi:10.14368/jdras.2021.37.2.61.